在本研究中,通过煅烧钴基普鲁士蓝类似物制备了Co3N纳米酶,用于活化PMS以降解吡虫啉。Co d 带中心的红移不仅增强了其对 PMS 的吸附能力,还通过调节 Co 的电子占据度优化了 Co 3d 与 O 2p 轨道之间的耦合,从而将反应路径从高能垒自由基途径转变为涉及直接生成 Co(IV)=O 的低能垒途径。与其他过渡金属基材料相比,该纳米酶在吡虫啉及其他有机污染物的氧化降解方面表现出优异的催化活性。吡虫啉的氧化降解活化能为 57.5 kJ/mol,远低于 Co-N2(115.6 kJ/mol)和 Co3O4(203.2 kJ/mol)。通过密度泛函理论(DFT)计算,阐明了Co₃N纳米酶的催化机理,以及Co(IV)=O物种的形成机制及其关键作用。氮的引入吸引了相连钴位点的电子,导致电子分布不均。DFT 计算表明,与 Co-N2 和 Co3O4 相比,氮掺杂使 Co3N 的 d 带中心更接近费米能级,并且 PMS 中钴物种与氧物种之间的电子耦合得到增强,从而加速了 O-O 键的断裂,并提高了 Co(IV)=O 的生成速率(方案 1)。
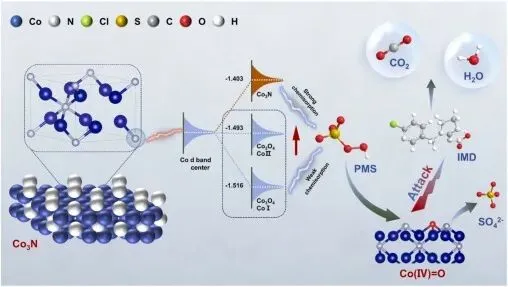
Scheme 1. Schematic diagram of the Co3N catalytic degradation mechanism.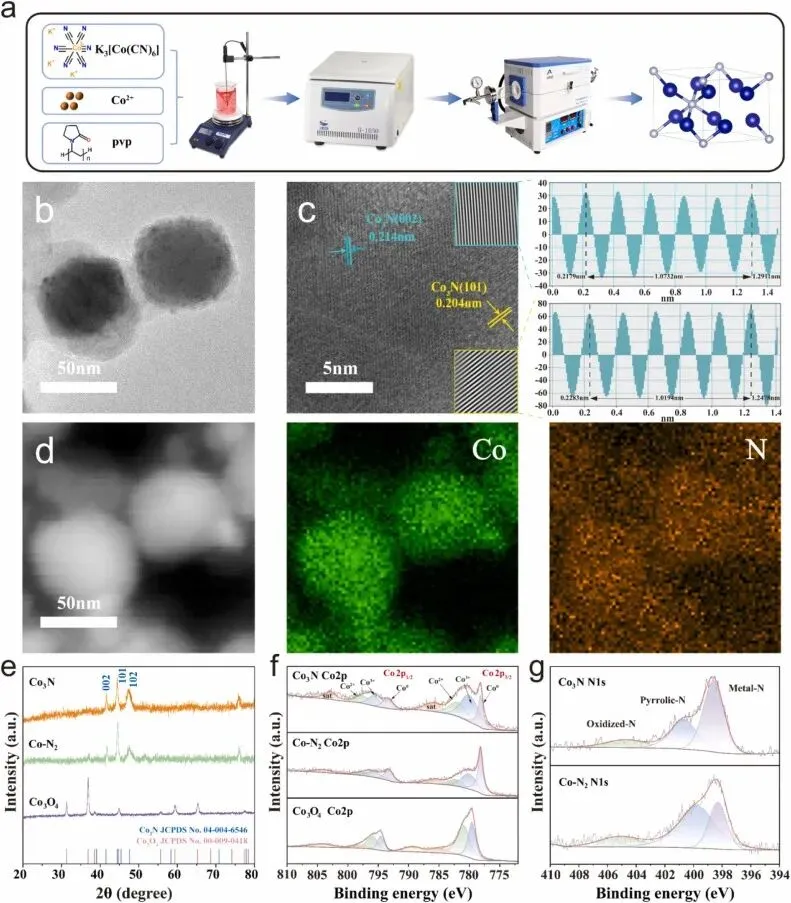
Fig. 1. (a) Schematic diagram of the synthesis strategy. (b) TEM of Co-MOF. (c) TEM images of Co3N. (d) HRTEM image and simulated lattice width of Co3N. (e) EDX elemental mappings of Co3N. (f) XRD images of Co3N, Co-N2 and Co3O4. (g) XPS survey spectra of Co in Co3N, Co-N2 and Co3O4. (h) XPS survey spectra of N in Co3N and Co-N2.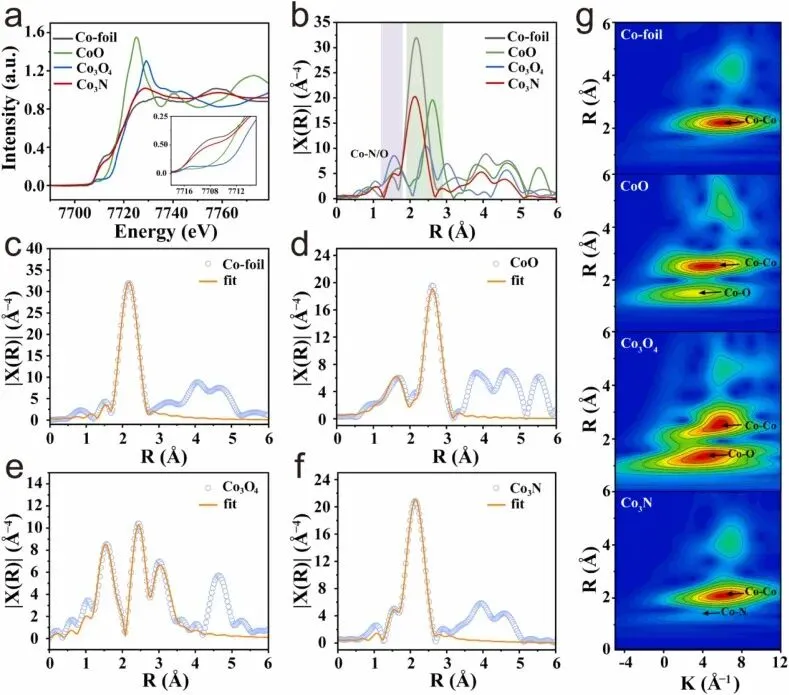
Fig. 2. (a) Normalization of Co K-edge XANES data for Co3N relative to standard samples. (b) R-space FT-EXAFS profiles of Co3N together with reference standards. (c-f) FT-EXAFS curve fitting of Co3N, Co foil, CoO, and Co3O4 in R space. (g) Wavelet transformation for the k3-weighted EXAFS signal of Co3N and reference samples.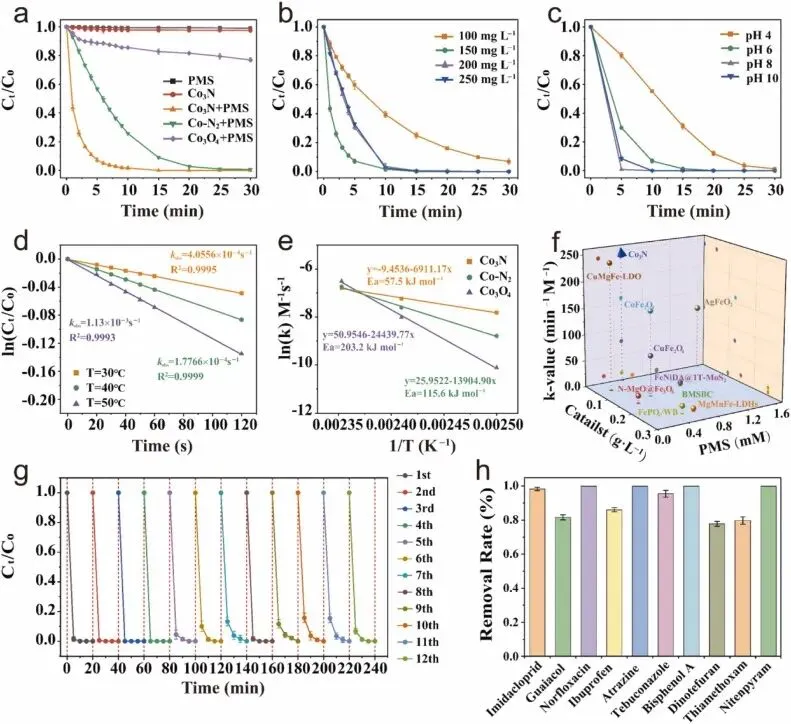
Fig. 3. Performance of the Co3N/PMS catalytic system. (a) Degradation of IMD under varied reaction conditions. (b) and (c) Degradation of IMD as a function of PMS concentration and pH in the Co3N /PMS process. (d) The corresponding reaction rate constants. (e) the Arrhenius plot over the Co3N. Reaction conditions: [pollutants] = 100 mg L−1. (f) Comparison of the Imidacloprid removal efficiency by Co3N with some reported catalysts. (g) IMD degradation in 12 consecutive cycles in the Co3N/PMS system. Reaction conditions: [catalyst] = 50 mg L−1, [pollutants] = 5 mg L−1. (h) Multiple pollutants were degraded within 10 min by the Co3N/PMS system. Reaction conditions: [catalyst] = 2 mg L−1, [PMS] = 150 mg L−1, [pollutants] = 10 mg L−1, [Temp.] = 25 ± 2 °C.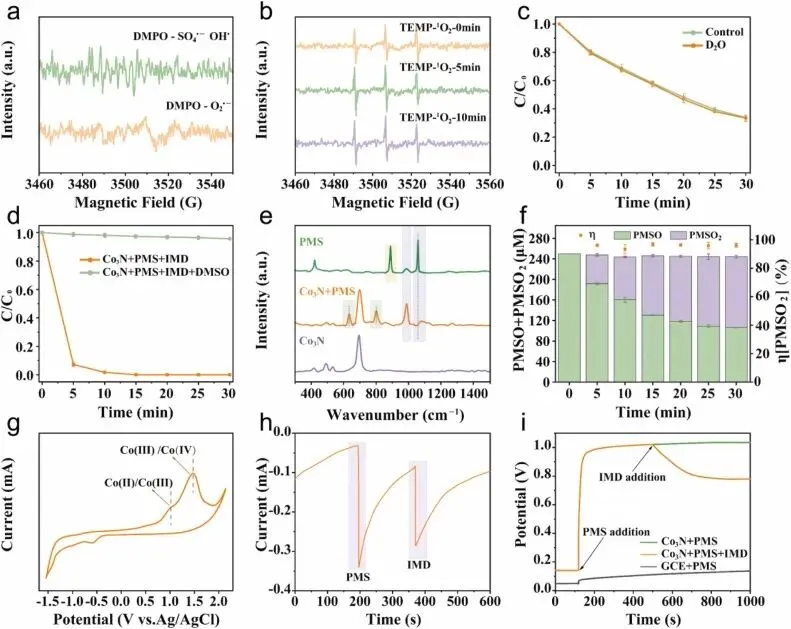
Fig. 4. (a), (b) EPR spectroscopy was employed to identify the ROS generated in the Co3N/PMS system. Reaction condition: [Co3N] = 0.2 g L−1, [PMS] = 1 mM, [Temp.] = 25 ± 2 °C, [Time] = 5 min−1. (c) Scavenging effect of D2O replacing H2O. (d) Degradation of Co3N/PMS in DMSO. (e) In situ Raman spectra of Co3N. (f) Variation in the total concentration of PMSO and PMSO2 along with the corresponding conversion efficiency η(PMSO2) during the reaction in the Co3N /PMS system. (g) Cyclic voltammetry (CV) measurements. (h) I-t of Co3N during PMS activation. (i) Chronopotentiometry (CP). Reaction conditions: [PMSO]0 = 250 μmol L−1, [Co3N]0 = 10 mg L−1.在本研究中,制备了过渡金属氮化物Co₃N纳米酶,该纳米酶在PMS介导的吡虫啉及其他有机污染物的氧化降解过程中表现出优异的催化活性。Co₃N/PMS体系中生成的Co(IV)=O可在15分钟内将吡虫啉(IMD)降解100%。Co₃N的归一化动力学速率常数(k)达到243.3 min⁻¹ M⁻¹,远高于已报道的催化剂。Co₃N具有良好的循环稳定性,经12次循环重复使用后仍保持超过95%的降解效率,并对多种类型的污染物表现出优异的降解性能。机理研究表明,Co₃N催化PMS降解有机污染物属于非自由基活化机制,其主要催化活性物种为Co(IV)=O。本研究提出了一种有前景的策略,通过同时克服“氧壁”限制并促进电子转移,有效增强了Co(IV)=O的生成。这种协同控制不仅显著提高了环境修复中的催化性能,还为设计先进的芬顿类催化剂奠定了合理的理论基础。
Minghua Wang, Yaxin Zheng, Dongchen Yang, Yichun Ren, Wei Gao, Ningzhao Shang, Jinlin Zhang, Shutao Gao, Zexiu An, Chun Wang, Fine modulating the D-band center of Co3N nanozyme for efficient oxidative degradation of emerging organic contaminants, Applied Catalysis B: Environment and Energy, 2026, https://doi.org/10.1016/j.apcatb.2026.126683
声明:本公众号仅分享前沿学术成果,无商业用途。如涉及侵权,请立刻联系公众号后台或发送邮件,我们将及时修改或删除!
邮箱:Environ2022@163.com